Difference between revisions of "PAA Selenium"
| Line 297: | Line 297: | ||
<math>\frac{{76 \atop 34\; }Se (\gamma,n){75 \atop \;Se}}{{80 \atop 34\; }Se (\gamma,n){79 \atop \;Se}} = \frac{Abundance \times \int_{10.02}^{18.02}\sigma dE}{Abundance \times \int_{10.02}^{18.02}\sigma dE} = \frac{0.0923 \times 2644.7}{0.498 \times 3090.5} = 0.16 = \frac{\sigma_{Se-75}}{\sigma_{Se-79}}</math> | <math>\frac{{76 \atop 34\; }Se (\gamma,n){75 \atop \;Se}}{{80 \atop 34\; }Se (\gamma,n){79 \atop \;Se}} = \frac{Abundance \times \int_{10.02}^{18.02}\sigma dE}{Abundance \times \int_{10.02}^{18.02}\sigma dE} = \frac{0.0923 \times 2644.7}{0.498 \times 3090.5} = 0.16 = \frac{\sigma_{Se-75}}{\sigma_{Se-79}}</math> | ||
| − | <math>\frac{{82 \atop 34\; }Se (\gamma,n){81 \atop \;Se}}{{80 \atop 34\; }Se (\gamma,n){79 \atop \;Se}} = \frac{Abundance \times \int_{10.02}^{18.02}\sigma dE}{Abundance \times \int_{10.02}^{18.02}\sigma dE} = \frac{0.0882 \times | + | <math>\frac{{82 \atop 34\; }Se (\gamma,n){81 \atop \;Se}}{{80 \atop 34\; }Se (\gamma,n){79 \atop \;Se}} = \frac{Abundance \times \int_{10.02}^{18.02}\sigma dE}{Abundance \times \int_{10.02}^{18.02}\sigma dE} = \frac{0.0882 \times 3271.3}{0.498 \times 3090.5} = 0.18</math> |
===Activity Productions=== | ===Activity Productions=== | ||
Revision as of 21:10, 12 January 2017
Using PAA ro measure Selenium concentrations.
According to Krouse<ref name="Krous1962"> H.R. Krause and H.G. Thode,"Thermodynamic Properties and Geochemistry of Iosotopic Compounds of Selenium",.Can. J. Chem., vol 40, pg 367</ref> , the fractional concentration of Se-82/Se-76 in plant material is observed to be less than from primordial (meteoric) concentrations by as much as 1.2%. Anaerobic bacteria are known to reduce selenates and senelites in biological systems. This may be the reason plant material has fractionation of selenium isotopes. They also observe excess concentrations of up to 0.4% in soil.
Plant material appears to detect environmental selenium.
Can one use plant material to measure the provenance of selenium?
Natural abundance of selenium
| Isotope | Abundance |
| Se-74 | 0.86% |
| Se-76 | 9.23% |
| Se-77 | 7.60% |
| Se-78 | 23.69% |
| Se-80 | 49.80% |
| Se-82 | 8.82% |
Possible reactions
| Reaction | Half-life | Relative activity | Gamma-rays, keV (BR) |
| Se-74(gamma,n)Se-73 | 7.1 h | 1.5E-1 | 361 (100) |
| Se-74(gamma,n)Se-73m | 39 m | 3.2 | 402 (4) |
| Se-74(gamma,np)As-72 | 26 h | 1.0E-3 | 834 (100) |
| Se-76(gamma,n)Se-75 | 120 d | 1.3E-2 | 265(29) |
| Se-77(gamma,p)As-76 | 26.4 h | 4.4E-2 | 559(44) |
| Se-78(gamma,p)As-77 | 38.8 h | 8.6E-2 | 239(2) |
| Se-80(gamma,n)Se-79m | 3.9 m | 5.9 | 96(10) |
| Se-80(gamma,np)As-78 | 1.5 h | 2.2E-2 | 614(54) |
| Se-80(gamma,p)As-79 | 8.2 m | 1.3 | 96(9) |
| Se-80(gamma,p)Ge-75 | 83 m | 2.8E-1 | 265(11) |
Can one perform PAA measurements of Se-82 and Se-76?
Neutron knockout of Se-82
If you knock a neutron out of Se-82 you produce the unstable isotope Se-81 which Beta emitts with half life of 18 min and a meta-state that emmits a 103 keV gamma with a 57 minute half life.
Other prominent photons
260 & 276 keV for the 57 minute half life isotope
The minimum energy required to knock out a neutron from this isotope is
(Mass of Se-81)-(Mass of Se-82)+(Mass of 1 neutron)[MeV/c^2] = 73511.651 - 744442.2136 + 939.57 -> 9.0 MeV
Neutron knockout of Se-76
If you knock a neutron out of Se-76 you produce the unstable isotope Se-75 which has a half life of 119 days.
The prominent photons emitted have the following energies
136, 264, and 279 keV
http://www.nucleide.org/DDEP_WG/Nuclides/Se-75_tables.pdf
The article below describes how plant material and soil contain Se-76 to Se-82 ratios that differ from other natural samples by 1.5%. They argue that it is due to the bacteria living in plant material.
File:Krouse CanJournChem 40 1962 p367.pdf
Plant material is a natural way to sample the selenium content to determine if there are difference isotopic ratios due to the impact of human activities on the environment.
The energy required to knock out a neutron from this isotope is
(Mass of Se-75) + (Mass of 1 neutron) - (Mass of Se-81)[MeV/c^2] = 69790.3321 + 939.57 - 70718.1 -> 11.8 MeV
Relative Yield Calculations
For this section, I am interested in finding the relative yield of Se-79 when compared to Se-81 and Se-75. Cross sections were found at this website: http://www-nds.indcentre.org.in/exfor/servlet/X4sSearch5?EntryID=220070
Cross-section
Below is a table of the cross sections for all of the reactions of interest (Se-76 -> Se-75, Se-82 -> Se-81, and Se-80 -> Se-79).
| Energy (MeV) | Se-80 -> Se-79 (mb) | Se-76 -> Se- 75 (mb) | Se-82 -> Se-81 (mb) |
| 9.42 | ** | ** | 5.58 +/- 0.5 |
| 9.62 | ** | ** | 8.7 +/- 0.5 |
| 9.82 | ** | ** | 10.8 +/- 0.5 |
| 10.02 | 7.1 +/- 0.2 | ** | 12.4 +/- 0.7 |
| 10.22 | 10.3 +/- 0.4 | 0.4 +/- 0.2 | 15 +/- 0.9 |
| 10.42 | 12.9 +/- 0.5 | 0.8 +/- 0.3 | 15.4 +/- 0.9 |
| 10.62 | 16.2 +/- 0.5 | 4.7 +/- 0.3 | 17.3 +/- 1.1 |
| 10.82 | 19.2 +/- 0.6 | 6.6 +/- 0.4 | 21 +/- 1.1 |
| 11.02 | 22 +/- 0.6 | 9.9 +/- 0.3 | 21.8 +/- 1.4 |
| 11.22 | 23.8 +/- 1 | 12.2 +/- 0.4 | 22.5 +/- 1.9 |
| 11.42 | 28 +/- 1 | 19.5 +/- 0.5 | 27.7 +/- 1.5 |
| 11.62 | 28.6 +/- 1.2 | 23.5 +/- 0.5 | 27.8 +/- 2 |
| 11.82 | 30.4 +/- 1.5 | 26.7 +/- 0.8 | 28.4 +/- 1.8 |
| 12.02 | 33 +/- 1.4 | 32 +/- 0.8 | 31.9 +/- 1.8 |
| 12.22 | 34.9 +/- 1.8 | 36.3 +/- 0.8 | 34.5 +/- 2.4 |
| 12.42 | 37.6 +/- 1.4 | 38.8 +/- 1.4 | 38.3 +/- 2.8 |
| 12.62 | 41.5 +/- 2 | 40.9 +/- 1.4 | 42.6 +/- 3 |
| 12.82 | 43.6 +/- 2.5 | 46.7 +/- 1.6 | 41 +/- 3.2 |
| 13.02 | 46.7 +/- 2.8 | 49.2 +/- 1.8 | 48.8 +/- 2.5 |
| 13.22 | 51.9 +/- 2.6 | 55.8 +/- 1.5 | 53.3 +/- 3.4 |
| 13.42 | 56.7 +/- 1.5 | 56 +/- 2.5 | 52.7 +/- 4.3 |
| 13.62 | 61.4 +/- 2.9 | 63.6 +/- 2.3 | 60.4 +/- 3.5 |
| 13.82 | 69.5 +/- 3.3 | 69.4 +/- 2.1 | 73.3 +/- 4.1 |
| 14.02 | 78.7 +/- 4.4 | 74.4 +/- 2.9 | 78.2 +/- 4.6 |
| 14.22 | 84.9 +/- 3.6 | 78 +/- 2.8 | 82.3 +/- 4.2 |
| 14.42 | 93.3 +/- 2.7 | 87.6 +/- 3.4 | 93 +/- 4.8 |
| 14.62 | 100.5 +/- 4 | 92.2 +/- 3.1 | 99.4 +/- 4.4 |
| 14.82 | 105.1 +/- 4.6 | 96.2 +/- 3 | 110.9 +/- 3.9 |
| 15.02 | 110.9 +/- 4.6 | 101.4 +/- 3.9 | 114.7 +/- 6.1 |
| 15.22 | 118.9 +/- 4.1 | 104.9 +/- 4.3 | 124.6 +/- 5.6 |
| 15.42 | 125.8 +/- 5.5 | 108.1 +/- 5.1 | 137.5 +/- 6.7 |
| 15.62 | 132.9 +/- 4.5 | 106.8 +/- 5 | 143.7 +/- 4.6 |
| 15.82 | 132.7 +/- 5.9 | 105.4 +/- 5.6 | 140.4 +/- 5 |
| 16.02 | 127.6 +/- 5.6 | 104.8 +/- 5 | 145.69 +/- 6.9 |
| 16.22 | 132.4 +/- 5.3 | 106.6 +/- 4.9 | 151.8 +/- 6.9 |
| 16.42 | 130 +/- 7.6 | 100.5 +/- 7.5 | 143 +/- 7.4 |
| 16.62 | 125.4 +/- 6.2 | 102.6 +/- 6.2 | 137.8 +/- 6.2 |
| 16.82 | 137.4 +/- 6.7 | 100.5 +/- 7.5 | 142.8 +/- 6 |
| 17.02 | 138.1 +/- 7.5 | 101 +/- 5.9 | 132.2 +/- 7.2 |
| 17.22 | 130.4 +/- 8.3 | 93.1 +/- 6.3 | 128.8 +/- 4.7 |
| 17.42 | 114.9 +/- 6 | 95.5 +/- 6.3 | 122.7 +/- 5.9 |
| 17.62 | 110.8 +/- 6 | 94.8 +/- 8 | 120.6 +/- 6.8 |
| 17.82 | 104.4 +/- 7.4 | 99.2 +/- 7.3 | 119.2 +/- 7.2 |
| 18.02 | 108.7 +/- 6.6 | 98.1 +/- 6.2 | 115.9 +/- 7 |
| 18.22 | 102.4 +/- 7.3 | 96.3 +/- 7.3 | 112.5 +/- 5.6 |
| 18.42 | 104.3 +/- 5.7 | 98.6 +/- 7.7 | 114.1 +/- 8.2 |
| 18.62 | 104.1 +/- 5.5 | 95.5 +/- 10 | 114.1 +/- 6.5 |
| 18.82 | 91.1 +/- 7.8 | 87.2 +/- 9.1 | 90.8 +/- 7.5 |
| 19.02 | 90.5 +/- 5.5 | 88.1 +/- 7.6 | 99.4 +/- 6.3 |
| 19.22 | 85.7 +/- 6.2 | 97 +/- 9.9 | 87.2 +/- 5.9 |
| 19.42 | 91.4 +/- 6.6 | 91.7 +/- 9.2 | 90.6 +/- 7.2 |
| 19.62 | 84.9 +/- 5.6 | 83.8 +/- 10 | 81.3 +/- 6.6 |
| 19.82 | 90.3 +/- 8.1 | 71.2 +/- 8 | 83.8 +/- 6.5 |
| 20.02 | 83 +/- 5.7 | 74.4 +/- 7.7 | 80 +/- 5.9 |
| 20.22 | 79.9 +/- 6.2 | 68.8 +/- 8 | 71.1 +/- 6.4 |
| 20.42 | 67.1 +/- 7.2 | 66.6 +/- 7 | 65 +/- 7.6 |
| 20.62 | 70.9 +/- 6.6 | 59.4 +/- 7.3 | 57 +/- 5.6 |
| 20.82 | 72.2 +/- 7.6 | 70.3 +/- 6.5 | 75.2 +/- 6.7 |
| 21.02 | 66.8 +/- 6.5 | 65.8 +/- 6.4 | 61.4 +/- 7.6 |
| 21.22 | 69.7 +/- 7 | 59.9 +/- 7.2 | 73.1 +/- 6.8 |
| 21.42 | 68.2 +/- 8.4 | 59.4 +/- 7 | 59 +/- 9.3 |
| 21.62 | 68.6 +/- 7.4 | 66.2 +/- 6.8 | 67.5 +/- 6.7 |
| 21.82 | 60.9 +/- 7.3 | 68 +/- 7.2 | 55 +/- 7.2 |
| 22.02 | 58.5 +/- 7.3 | 62.1 +/- 6.5 | 55.9 +/- 8.6 |
| 22.22 | 56 +/- 7.8 | 65.3 +/- 7 | 59.4 +/- 8.1 |
| 22.42 | 69.7 +/- 7.7 | 61.6 +/- 0.4 | 64 +/- 9.7 |
| 22.62 | 60.1 +/- 8.8 | 67.7 +/- 6.2 | 67 +/- 8.2 |
| 22.82 | 60.9 +/- 8.7 | 55.7 +/- 7.8 | 55.9 +/- 8.6 |
| 23.02 | 58.8 +/- 9.3 | 57.2 +/- 6.1 | 61.4 +/- 11.2 |
| 23.22 | 50.6 +/- 9.7 | 55.2 +/- 7.7 | 48.5 +/- 8.1 |
| 23.42 | 31.5 +/- 8.7 | 44.7 +/- 8.1 | 46.5 +/- 10.1 |
| 23.62 | 37.7 +/- 8.7 | 31.3 +/- 5.9 | 34 +/- 7.1 |
| 23.82 | 29.5 +/- 10.2 | 34.7 +/- 5.9 | 47.6 +/- 10.1 |
| 24.02 | 32.4 +/- 8.6 | 33 +/- 6.3 | 43.4 +/- 10.6 |
| 24.22 | 32.8 +/- 9.6 | 21.7 +/- 7.3 | 46.2 +/- 9.5 |
Similar data can be found in the link above. Below is the plot for all the reactions of interest.
Below is a plot of 3 different isotopes of selenium with their cross sections as a function of energy.
Now to find the relative yields, I approximated the integral the above graph using a left hand Riemann sum with an interval width of 1 MeV and multiplied it by the natural abundance in the sample. Below is a table of the reactions and their integrated cross sections.
| Reaction | Abundance | Integrated Cross Section (mb) [10->24 MeV] |
| Se-82(gamma,n)Se-81 | 8.82% | 5429.19 |
| Se-80(gamma,n)Se-79 | 49.8% | 5249.6 |
| Se-76(gamma,n)Se-75 | 9.23% | 4703.1 |
| Reaction | Abundance | Integrated Cross Section (mb) [10->18 MeV] |
| Se-82(gamma,n)Se-81 | 8.82% | 3271.3 |
| Se-80(gamma,n)Se-79 | 49.8% | 3090.5 |
| Se-76(gamma,n)Se-75 | 9.23% | 2644.7 |
Below are the equations for the relative yield of each isotope.
As of recently, the beam energy has been lowered to 18 MeV, so the relative yield for each isotope now becomes
Activity Productions
Let
- = The number of activated atoms per cubic cm at time
- =The number density of the target = [ Atoms/cm^3]
- = material density [g/cm^3]
- = Avagadro's number = g/mole
- = Atomic Number
- = incident photon flux ( photons/sec/cm/cm)
The rate of activated nuclei production is estimated by subtracting the rate of decay from the rate of production
where
= half life of activated nucleus
The production rate ratio of Se-79 to Se-75 is
Experiments
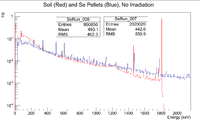
Preliminary Information Before Irradiation of Soil Sample
Using Detector D at the IAC, I had to get a count on the unirradiated soil sample that was collected. Here are the GPS coordinates ****
Here is the spectrum produced:
The details of the calibration along with the constants found from a linear fit of temperature vs. voltage can be found in the page below.
Calibration 10-21-16 Det D Calibration 10-28-16 Det A
Nickel Normalization
Talk about Nickel reaction, give example of Nickel rates for 511 and 1377 keV lines. Run condition for examples below.
Using a 45 MeV linac (Jack) you get 75 uCi of Nickel per (gram Kw hr)
If using a 25 meV machine at 1 kW power, how ling should I run to keep 1 g of natural Selium from having a Se-79 Activiy less than 16 pCi?
Here is the data for the cross section of Ni-58(,n)Ni-57 data
http://www-nds.indcentre.org.in/exfor/servlet/X4sGetSubent?reqx=119235&subID=220597006&plus=1
When comparing this data to the Se-80(,n)Se-79 reaction, we must be careful when handling the bin sizes. The data for the Se-79 reaction is binned by intervals of 0.2 MeV, while the Ni-57 data is binned by intervals of 0.1 MeV. To create coarser bins, I must take the 3 data points from the Nickel data and get an average value, then plot that value centered at the midpoint of these two values. For example, I would take 10.1,10.2, and 10.3 MeV and average their values and plot at 10.2 MeV. Below is the plot:
Here is the data for the plot above. The Zn-68 -> Cu-67 data was found in the IAEA handbook on page 160. I used data thief to find the points that were plotted.
| Energy (MeV) | Zn-68 -> Cu-67 (mb) | Ni-58 -> Ni-57 | Se-80 -> Se-79 |
| 12.0 | 0.0103 | ** | 33.8 +/- 1.8 |
| 12.2 | 0.0117 | 0.56 +/- 0.2 | 35.6 +/- 1.7 |
| 12.4 | 0.0128 | 1.56 +/- 0.2 | 40.8 +/- 1.9 |
| 12.6 | 0.0624 | 2.83 +/- 0.2 | 42.3 +/- 1.5 |
| 12.8 | 0.1496 | 4.23 +/- 0.2 | 47.4 +/- 2.2 |
| 13.0 | 0.2113 | 5.13 +/- 0.2 | 51.6 +/- 2.8 |
| 13.2 | 0.3149 | 5.53 +/- 0.2 | 54.8 +/- 2.6 |
| 13.4 | 0.5009 | 5.93 +/- 0.2 | 61.9 +/- 2.8 |
| 13.6 | 0.642 | 6.6 +/- 0.2 | 69.5 +/- 2.4 |
| 13.8 | 0.7151 | 7.46 +/- 0.2 | 74.4 +/- 3.3 |
| 14.0 | 0.8874 | 8.79 +/- 0.2 | 82.8 +/- 3.8 |
| 14.2 | 0.9466 | 9.87 +/- 0.2 | 94.1 +/- 3.4 |
| 14.4 | 0.9884 | 10.41 +/- 0.2 | 94 +/- 3.6 |
| 14.6 | 1.113 | 10.47 +/- 0.2 | 102 +/- 3.4 |
| 14.8 | 1.337 | 10.92 +/- 0.2 | 107.3 +/- 4.3 |
| 15.0 | 1.411 | 11.71 +/- 0.2 | 111.3 +/- 4.7 |
| 15.2 | 1.4574 | 12.76 +/- 0.2 | 115.6 +/- 4.7 |
| 15.4 | 1.6059 | 14.09 +/- 0.2 | 119.9 +/- 5 |
| 15.6 | 1.641 | 15.76 +/- 0.2 | 120.1 +/- 5.3 |
| 15.8 | 1.7318 | 18.13 +/- 0.2 | 126 +/- 7.3 |
| 16.0 | 1.7506 | 21.22 +/- 0.2 | 122.9 +/- 7.3 |
| 16.2 | 2.0141 | 22.89 +/- 0.2 | 114.6 +/- 5 |
| 16.4 | 2.2435 | 23.13 +/- 0.2 | 122.4 +/- 6.1 |
| 16.6 | 2.3172 | 22.84 +/- 0.2 | 121.2 +/- 7.7 |
| 16.8 | 2.6092 | 22.61 +/- 0.2 | 118.2 +/- 9.1 |
| 17.0 | 2.6947 | 22.31 +/- 0.22 | 116.8 +/- 8.8 |
| 17.2 | 2.9378 | 22.86 +/- 0.39 | 116.4 +/- 5.9 |
| 17.4 | 3.1341 | 24.23 +/- 0.46 | 109.5 +/- 7.4 |
| 17.6 | 3.3798 | 25.8 +/- 0.54 | 109.6 +/- 8.3 |
| 17.8 | 3.2368 | 27.12 +/- 0.63 | 102.3 +/- 8.1 |
| 18.0 | 4.0164 | 27.38 +/- 0.63 | 107.2 +/- 8.3 |
| 18.2 | 4.4739 | 26.5 +/- 0.62 | 111.9 +/- 8 |
| 18.4 | 4.5713 | 25.18 +/- 0.64 | 101.3 +/- 8.3 |
| 18.6 | 4.6206 | 24.16 +/- 0.64 | 97 +/- 7.5 |
| 18.8 | 5.1468 | 23.31 +/- 0.71 | 93.4 +/- 7.9 |
| 19.0 | 5.0914 | 22.38 +/- 1.10 | 90.5 +/- 7.1 |
| 19.2 | 5.6712 | 20.84 +/- 1.71 | 87.1 +/- 7.4 |
| 19.4 | 6.525 | 19.18 +/- 1.51 | 89.7 +/- 7.8 |
| 19.6 | 6.8866 | 19.93 +/- 1.10 | 90.6 +/- 6.3 |
| 19.8 | 6.9607 | 24.06 +/- 1.3 | 90.3 +/- 6.3 |
| 20.0 | 7.6705 | 29.25 +/- 0.9 | 84.9 +/- 7 |
| 20.2 | 8.1832 | 31.17 +/- 0.69 | 79 +/- 6.2 |
| 20.4 | 8.4519 | 29.6 +/- 0.69 | 75.5 +/- 7.9 |
| 20.6 | 8.8241 | 27.06 +/- 0.87 | 74.1 +/- 6.5 |
| 20.8 | 9.5163 | 26.29 +/- 0.82 | 75.4 +/- 6.4 |
| 21.0 | 9.9354 | 25.4 +/- 0.72 | 72.9 +/- 7.4 |
| 21.2 | 10.0428 | 23.07 +/- 0.76 | 67.6 +/- 5.6 |
| 21.4 | 10.151 | 21.84 +/- 0.84 | 69.1 +/- 8.3 |
| 21.6 | 10.1502 | 23.90 +/- 0.79 | 57.2 +/- 7.7 |
| 21.8 | 10.149 | 25.98 +/- 0.88 | 61 +/- 7.6 |
| 22.0 | 9.9323 | 25.64 +/- 1.27 | 58 +/- 7.6 |
| 22.2 | 9.9316 | 21.39 +/- 2.20 | 57.8 +/- 6.4 |
| 22.4 | 9.7191 | 17.49 +/- 1.49 | 65.8 +/- 9.6 |
| 22.6 | 9.3077 | 15.82 +/- 0.82 | 59.9 +/- 6.9 |
| 22.8 | 8.9141 | 15.85 +/- 0.76 | 55.7 +/- 8.4 |
| 23.0 | 8.7229 | 16.79 +/- 1.10 | 53.4 +/- 7.3 |
| 23.2 | 8.6287 | 17.07 +/- 0.86 | 57.5 +/- 7.8 |
| 23.4 | 8.2638 | 15.93 +/- 0.93 | 32 +/- 7.8 |
| 23.6 | 8.0867 | 12.61 +/- 0.84 | 43.7 +/- 7.8 |
| 23.8 | 7.8288 | 8.76 +/- 0.87 | 36.5 +/- 9.8 |
| 24.0 | 7.5787 | 6.96 +/- 0.78 | 39.7 +/- 7.2 |
| 24.2 | 7.8 | 6.45 +/- 0.86 | 37.3 +/- 8.5 |
Using a left handed Riemann Sum on each set of cross section data (The Selenium 79 reaction and the Nickel 58 reaction), the relative activity between the nickel isotopes and the selenium isotopes can be found. For the Nickel reaction I used a rectangular width equal to the bin size of the measurements, which was 0.1 MeV. Similarly for the selenium I had to use a bin size of 0.2 MeV while rounding the values down to the nearest tenth, i.e. 10.02 becomes 10.0, 10.22 becomes 10.2 and so on.
| Reaction | Integrated Cross Section (mb) | Abundance |
| Se-80(gamma,n)Se-79 | 5429.6 | 49.8% |
| Ni-58(gamma,n)Ni-57 | 214.047 | 68.1% |
| Zn-68(gamma,p)Cu-67 | 293.2027 | 18.45 % |
Here Segebade has the inverse of this expression, so taking the inverse we have my 0.53 compared to Segebade's 0.54
Here Segebade has the inverse of this expression, so taking the inverse we have my 0.45 compared to Segebade's 0.013
= 0.000000059
Now using the production rate ratio above from the nickel we can determine that the production rate of Se-79 is 4.425pCi/h, this would mean that we could irradiate for 3.6 hours before reaching the threshold of 16pCi.
Segebade has a table of ratios of specific activities of certain elements compared to Ni-57. The section starts on page 167, but the ratios with selenium are found on page 176
Since the beam energy has been changed to 18 MeV, I will recalculate the expected yield with respect to nickel
= 0.000000066
Now 75 microCi/h (assuming 1 kW and 1 g of natural selenium) 75(microCi/h)*0.000000066 = 0.000004949 microCi/h = 4.9 pCi/h, so we can irradiate for 3.27 hours before we hit 16 pCi.
Chlorine
It looks like Cl-35 is abundant as you see photon energies of 146 keV and 2127 keV (you can barely see 1176 keV) from Cl-34's decay (neutron knocked out of Cl-35).
The half life is 32 minutes.
Should check the half life from the run AccOnAlInDetASe-AinDetD_001.root using the calibration
MPA->Draw("0.18063+0.960133*evt.Chan>> SeRun_008(8000,0.5,8000.5)","evt.ADCid==3");
Irradiation of Horse Mineral Supplement
Below is the EMSL report for the horse feed sample. https://wiki.iac.isu.edu/index.php/File:EMSL_Report_Horse_Feed.pdf
Chlorine is a dominant signal
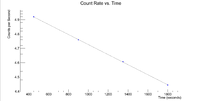
First, look at the peak around 146 keV
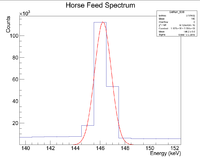
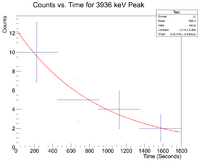
Next I plotted the counts as a function of time to get an exponentially decaying graph. When doing an exponential fit here, the parameter "b" given by root will be the decay constant.
Root gives a half life of 32.9508 +/- 0.01 minutes
Now do the same for the 2127 keV line
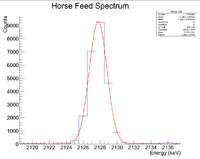
Here are the counts plotted as a function of time
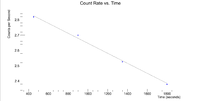
Root gives a half life of 35.3962 +/- 0.2 minutes
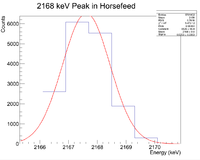
Potassium is a potential signal
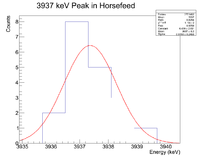
Looking at the spectrum for the fast irradiation sample, there are 2 prominent lines that could be from 38-K. The mechanism would be a single neutron knockout from a stable 39-K nucleus. The two most dominant energies of the three for 38-K are 2167 keV and 3936 keV and the half life is 7.63 minutes. Below is a fit to the energy spectrum histogram
Now check the half life
Root gives a half life of 8.03 +/- 0.02 minutes
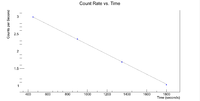
Next check the 3936 peak
and check the half life
Root gives a value for b = - 1.14372x10^(-3), which in turn gives a half life of 10.1 minutes
It seems very possible that 38-K could be in the sample of horse feed.
First Observation of Se lines
Using the 44 Machine at 7 kW power and 44 meV incident electron energy to produce a bremsstrahlung spectrum with a mean energy of 15 meV.
All runs lasting less than 214 seconds have time stamp that gives real time if you divide by clock frequency of 20 MHz. The first 32 bits are used for a real time measurement.
MDA and Se mass Calculations
To find the mass of the selenium in the irradiated horse feed sample, we need some masses and volumes of the horse feed. A vial containing 20mL of regular horse feed was massed. The mass of the vial was 13.6406g and the total mass of the vial and the horse feed was 31.8504g. This means the mass of the horse feed is . Since this mass is in a 20mL = 0.02L container, the density of the horse feed (assuming the mass difference between the non-irradiated and the irradiated horse feed is negligible) is
.
Now for the irradiated horse feed the mass of the container is 25.0259g and the total mass of the container and the sample is 43.7529g. The mass of the sample then is 43.7529g - 25.0259g = 18.727g. Now using the density found before we can find the volume of the irradiated horse feed within the container.
The EMSL report says that there are in the horse feed, so the mass of the selenium in the horse feed sample is
.
Each of the calibration runs were used at position C in detector D. Below is the runlist for the isotopes used. The intervals used were (117:127) and (272:282) respectively.
| Source | Serial # | Reference Date | Activity | Start | Stop | Live |
| Co-57 | 129735 | 07/01/08 | 1.074 micro Ci | 16:17:37 | 16:27 | 601.10 |
| Ba-133 | 129790 | 07/01/08 | 1.188 micro Ci | 16:29:14 | 16:32 | 208.259 |
For the Co-57, the activity as of 09/07/16 is 0.0005 microCi and the intensity of the 122.3 keV line is 85.6%
- = for the 122.3 line
For the Ba-133, the activity as of 09/07/16 is 0.69 microCi and the intensity of the 277.05 keV line is 7.164%
- = for the 277.05 line
| Run | Source | Energy (keV) | Expected Rate (Hz) | HpGe Rate (Hz) | HpGe Det D Efficiency (%) |
| Eff_C_Co_57.root | Co-57 | 122.3 | 15.84 | 0.4526 - 0.2144 | 1.5 |
| Eff_C_Ba_133.root | Ba-133 | 277.05 | 1828.97 | 20.58 - 0.07641 | 1.12 |
Now that the efficiency has been found, we must find the background rate to find the MDA. To find the background rate I used ROOT and plotted the energy spectra for Se_B_005 and HorseFeed_NoIrr. The windows of interest are the same as above. Each run's time was cut down to 1 hour.
First I will find the MDA for the window from (117:127). In Se_B_005, the activity was found to be 0.8433 HZ and in the HorseFeed_NoIrr the activity was found to be 0.2133 Hz, which gives a background rate of 0.63 Hz -> 37.6 cpm.
Now we can compute the MDA using this reference https://wiki.iac.isu.edu/index.php/File:Nwsltr-43re.pdf
I found the MDA to be [(2.71 + 4.65*(37.6 * 60)^1/2]/(60*0.015) = 249.1 dpm
Now I will find the MDA for the window from (272:282). In Se_B_005, the activity was found to be 0.7408 Hz and in HorseFeed_NoIrr the activity was found to be 0.08278. This means that the background activity is 0.66 Hz -> 39.48 cpm.
Now we can compute the MDA = [(2.71 + 4.65*(39.48 * 60)^1/2]/(60*0.012) = 318.1 dpm
Detector Efficiency
Below is the runlist for finding the efficiency of the detector at position R
| Source | Serial # | Reference Date | Activity | Start | Stop | Live |
| Na-22 | 129743 | 7-01-08 | 9.427 microCi | 15:49 | 16:19 | 1796.803 |
| Cs-137 | 129793 | 7-01-08 | 1.006 microCi | 14:25 | 14:56 | 1879.606 |
| Mn-54 | 129807 | 7-01-08 | 11.77 microCi | 15:00 | 15:30 | 1793.420 |
| Co-60 | 129740 | 7-01-08 | 10.42 microCi | 15:33 | 15:43 | 569.725 |
Below are the theoretical calculations for the theoretical decay frequencies
Na-22, 9.427micro Ci on July 1, 2008, half life 2.602 +/- 0.002 years, 99.937% for 1274.52 and 178.8 for 511 line , activity in March 31, 2016 =1.196micro Ci
- = for the 1274 line
- = for the 511 line
Cs-137, 661.660 line, 85.21% * 1.066micro Ci on July 1, 2008, half life 30.0 +/- 0.2 yrs, March 31, 2016 activity = 0.891micro Ci expected rate for 661 line
- =
Mn-54, 11.77 microCi on July 1, 2008, half life =312.20 +/- 0.07 days, 99.975% intensity on 834.826 , March 31, 2016 activity =0.02328micro Ci
- = for the 834 line
Co-60, 10.42micro Ci July 1, 2008, half life 5.271 +/- 0.001 years, 99.0 % for 1173.237 and 99.9824 % for 1332.501, March 31, 2016 activity=3.759micro Ci
- = for the 1173 line
- = for the 1332 line
Below is a table where the actual efficiency will be calculated for position R (farthest position).
| Run | Source | Energy (keV) | Expected Rate (Hz) | HpGe Rate (Hz) | HpGe Det D Efficiency (%) |
| Eff_003 | Na-22 | 511 | 79122.6 | (506:516) (4.309-0.065=4.244) | 0.005 |
| Eff_005 | Cs-137 | 661.657 | 28091.2 | (657:666)(1.105-0.02281=1.0821) | 0.004 |
| Eff_006 | Mn-54 | 834.848 | 861.1 | (830:839)(0.04037-0.009123=0.031247) | 0.004 |
| Eff_007 | Co-60 | 1173.228 | 137692 | (1164:1182)(3.686-0.01939=3.67) | 0.003 |
| Eff_003 | Na-22 | 1274.537 | 44224 | (1270:1279) (1.073-0.0057=1.0673) | 0.002 |
| Eff_007 | Co-60 | 1332.492 | 139058.52 | (1328:1337)(3.283-0.05702=3.22598) | 0.002 |
Below is a runlist for position k
| Source | Serial # | Reference Date | Activity | Start | Stop | Live |
| Na-22 | 129743 | 7-01-08 | 9.427 microCi | 14:54 | 15:01 | 434.087 |
| Cs-137 | 129793 | 7-01-08 | 1.006 microCi | 15:48 | 15:55 | 413.925 |
| Mn-54 | 129807 | 7-01-08 | 11.77 microCi | 15:28 | 15:40 | 705.186 |
| Co-60 | 129740 | 7-01-08 | 10.42 microCi | 15:41 | 15:47 | 346.092 |
Below are the theoretical decay frequencies
Na-22, 9.427micro Ci on July 1, 2008, half life 2.602 +/- 0.002 years, 99.937% for 1274.52 and 178.8 for 511 line , activity in April 14, 2016 =1.183micro Ci
- = for the 1274 line
- = for the 511 line
Cs-137, 661.660 line, 85.21% * 1.066micro Ci on July 1, 2008, half life 30.0 +/- 0.2 yrs, April 14, 2016 activity =0.890 micro Ci expected rate for 661 line
- =
Mn-54, 11.77 on July 1, 2008, half life =312.20 +/- 0.07 days, 99.975% intensity on 834.826 , April 14, 2016 activity =0.02251micro Ci
- = for the 834 line
Co-60, 10.42micro Ci July 1, 2008, half life 5.271 +/- 0.001 years, 99.0 % for 1173.237 and 99.9824 % for 1332.501, April 14, 2016 activity=3.74micro Ci
- = for the 1173 line
- = for the 1332 line
Below are the actual efficiencies for position k
| Run | Source | Energy (keV) | Expected Rate (Hz) | HpGe Rate (Hz) | HpGe Det D Efficiency (%) |
| Eff_k_002 | Na-22 | 511 | 79122.6 | (506:516)(49.21-0.6272=48.58) | 0.06 |
| Eff_k_006 | Cs-137 | 661.657 | 28091.2 | (657:666)(12.86-0.02281=12.837) | 0.05 |
| Eff_k_004 | Mn-54 | 834.848 | 861.1 | (830:839)(0.3204-0.009123=0.311) | 0.04 |
| Eff_k_005 | Co-60 | 1173.228 | 137692 | (1164:1182)(42.39-0.02053=42.369) | 0.03 |
| Eff_k_002 | Na-22 | 1274.537 | 44224 | (1270:1279) (12.53-0.005702)=12.52 | 0.03 |
| Eff_k_005 | Co-60 | 1332.492 | 139058.52 | (1328:1337)(35.94-0.005072) | 0.03 |
Below is a runlist for position C
| Source | Serial # | Reference Date | Activity | Start | Stop | Live |
| Na-22 | 129742 | 7-01-08 | 1.146 microCi | 12:55 | 12:57 | 129.782 |
| Cs-137 | 129793 | 7-01-08 | 1.006 microCi | 13:02 | 13:04 | 123.818 |
| Mn-54 | 129806 | 7-01-08 | 1.226 microCi | 13:11 | 13:21 | 613.754 |
| Co-60 | 129739 | 7-01-08 | 1.082 microCi | 13:08 | 13:09 | 103.599 |
Below are the calculations for the theoretical frequencies
Na-22, 9.427micro Ci on July 1, 2008, half life 2.602 +/- 0.002 years, 99.937% for 1274.52 and 178.8 for 511 line , activity in May 5, 2016 =1.196micro Ci
- = for the 1274 line
- = for the 511 line
Cs-137, 661.660 line, 85.21% * 1.066micro Ci on July 1, 2008, half life 30.0 +/- 0.2 yrs, May 5, 2016 activity = 0.89micro Ci expected rate for 661 line
- =
Mn-54, 1.226 microCi on July 1, 2008, half life =312.20 +/- 0.07 days, 99.975% intensity on 834.826 , May 5, 2016 activity =0.002micro Ci
- = for the 834 line
Co-60, 1.082micro Ci July 1, 2008, half life 5.271 +/- 0.001 years, 99.0 % for 1173.237 and 99.9824 % for 1332.501, May 5, 2016 activity=0.39micro Ci
- = for the 1173 line
- = for the 1332 line
Below is a table with the calculated efficiencies
| Run | Source | Energy (keV) | Expected Rate (Hz) | HpGe Rate (Hz) | HpGe Det D Efficiency (%) |
| Eff_C_001 | Na-22 | 511 | 9261.8 | (506:516) (45.65-0.065=45.585) | 0.5 |
| Eff_C_002 | Cs-137 | 661.657 | 28059.7 | (657:666)(101.7-0.02281=101.67) | 0.4 |
| Eff_C_004 | Mn-54 | 834.848 | 73.98 | (830:839)(0.2704-0.009123=0.2612) | 0.4 |
| Eff_C_003 | Co-60 | 1173 | 14285.7 | (1164:1182)(34.24-0.01939=34.22) | 0.2 |
| Eff_C_001 | Na-22 | 1274.537 | 5176.7 | (1270:1279) (11.15-0.0057=11.14) | 0.2 |
| Eff_C_003 | Co-60 | 1332.492 | 14427.5 | (1328:1337)(28.05-0.05702=27.99) | 0.2 |
Run List
| Date | Time elapsed (Seconds) | Sample | Document Title | Start | Stop | Real | Live | Position |
| 04-01-16 | 2.16x10^6 | Se_B | Se_B_002 | 15:55 | 09:15 | 235989.882 | 235687.660 | k |
| 04-06-16 | 2.592x10^6 | Se_B | Se_B_003 | 12:57 | Interrupted | computer | crash | k |
| 04-14-16 | 3.283x10^6 | Se_B | Se_B_005 | 15:57 | 09:37 | 63581.784 | 63509.895 | k |
| 04-15-16 | 3.37x10^6 | Sample D | Sample_D_001 | 14:47 | 08:23 | 236172.264 | 236173.271 | k |
| 04-19-16 | 3.715x10^6 | Sample B | Sample_B_001 | 15:31 | 15:18 | 85634.862 | 85624.090 | k |
| 4-20-16 | 3.802x10^6 | Sample C | Sample_C_001 | 15:22 | 10:19 | 68253.774 | 68232.238 | k |
| 04-21-16 | 3.888x10^6 | Sample A | Sample_A_001 | 10:22 | 10:37 | 87292.409 | 87268.114 | k |
| 04-25-16 | 4.234x10^6 | Sample E | Sample_E_001 | 11:36 | 10:03 | 80822.406 | 80795.679 | k |
| 04-26-16 | 4.32x10^6 | Se_B | Se_B_008 | 10:06 | 10:29 | 87784.755 | 87664.070 | k |
| 05-05-16 | 5.098x10^6 | Sample A | Sample_A_002 | 13:31 | 14:30 | 3605.507 | 3602.925 | c |
| 05-05-16 | 5.098x10^6 | Sample B | Sample_B_002 | 14:34 | 15:26 | 3114.244 | 3112.620 | c |
| 05-05-16 | 5.098x10^6 | Sample C | Sample_C_002 | 15:28 | 10:57 | 70124.788 | 70044.470 | c |
| 05-06-16 | 5.184x10^6 | Sample D | Sample_D_002 | 10:59 | 15:34 | 16516.898 | 16512.570 | c |
| 05-06-16 | 5.184x10^6 | Sample E | Sample_E_004 | 15:37 | 16:18 | 261654.225 | 261344.308 | c |
| 05-09-16 | 5.443x10^6 | Se B | Se_B_012 | 16:20 | 11:08 | 67157.101 | 66660.298 | c |
| 05-10-16 | 5.5296x10^6 | Sample A | Sample_A_004 | 11:03 | 15:19 | 15379.475 | 15363.017 | c |
| 05-10-16 | 5.5296x10^6 | Sample B | Sample_B_004 | 15:22:04 | 11:43 | 73256.181 | 73220.324 | c |
| 05-16-16 | 6.048x10^6 | Sample C | Sample_C_004 | 16:33 | 08:19 | 56758.980 | 56711.121 | c |
| 05-18-16 | 6.2208x10^6 | Sample D | Sample_D_006 | 08:44:21 | 14:05 | 19271.829 | 19266.929 | c |
| 05-18-16 | 6.2208x10^6 | Sample E | Sample_E_006 | 14:08 | 08:06 | 151108.258 | 150955.915 | c |
| 05-20-16 | 6.3936x10^6 | Se_B | Se_B_014 | 08:08:47 | 08:44 | 261353.204 | 259621.655 | c |
| 05-23-16 | 6.6528x10^6 | Sample A | Sample_A_006 | 08:48 | 13:49 | 18103.004 | 18091.523 | c |
| 05-23-16 | 6.6528x10^6 | Sample B | Sample_B_006 | 13:52 | 13:24 | 84763.938 | 84696.083 | c |
| 05-24-16 | 6.7392x10^6 | Sample C | Sample_C_006 | 13:28:28 | 10:28 | 75571.716 | 75502.871 | c |
| 05-31-16 | 7.344x10^6 | Sample B | Sample_B_008 | 08:57:22 | 08:55 | 86282.861 | 86237.392 | c |
| 06-01-16 | 7.4304x10^6 | Sample C | Sample_C_008 | 08:58:39 | 13:31 | 102739.504 | 102647.471 | c |
| 06-02-16 | 7.5168x10^6 | Sample D | Sample_D_010 | 13:33 | 08:41 | 68915.044 | 68898.246 | c |
References
<references/>
File:Krouse CanJournChem 40 1962 p367.pdf
Goryachev, A. M., & Zalesnyy, G. N. (n.d.). The studying of the photoneutron reactions cross sections in the region of the giant dipole resonance in zinc, germanium, selenium, and strontium isotopes. Retrieved September 16, 2016, from http://www-nds.indcentre.org.in/exfor/servlet/X4sSearch5?EntryID=220070
Goryachev, B. I., Ishkhanov, B. S., Kapitonov, I. M., Piskarev, I. M., Piskarev, V. G., & Piskarev, O. P. (n.d.). Giant Dipole Resonance on Ni Isotopes. Retrieved October 26, 2016, from http://www-nds.indcentre.org.in/exfor/servlet/X4sGetSubent?reqx=119235&subID=220597006&plus=1
Handbook on Photonuclear data for applications, cross sections, and spectra. (2000, October). Retrieved November 4, 2016, from http://www-pub.iaea.org/MTCD/Publications/PDF/te_1178_prn.pdf
MSDS
Selenium shot, amorphous, 2-6 mm, Puratronic, 99.999% Alfa Aesar product # 10603 File:AlphaAesarSelenium MDSD.pdf
Informative links
https://inldigitallibrary.inl.gov/sti/3169894.pdf