Difference between revisions of "Induced Neutron Fission Fragment"
| Line 1: | Line 1: | ||
= Neutron Emission= | = Neutron Emission= | ||
| − | They are two typs of neutron sources: Natural neutron sources, and | + | They are two typs of neutron sources: Natural neutron sources, and man-made neutron sources. |
Revision as of 22:24, 23 May 2013
Neutron Emission
They are two typs of neutron sources: Natural neutron sources, and man-made neutron sources.
- A-Natural Neutron Sources (Intrinsic Sources)
These type of sources are naturally exits, they mainly emit neutrons by spontaneous fission. An examples of they sources are listed in table below.
Some of these sources are created by nuclear reaction in a reactor as it will be discussed below.
| Nuclide | Fission | -decay | Neutron/sec.gram |
| years | years | ||
| years | years | ||
| years | years | ||
| years | years | ||
| years | years | ||
| 66 years | 2.65 years |
- Cf-252
Cl-252 is one of the most commonly used natural source in industry and laboratories. Cf-252 has a continuous neutron spectrum up to 10 MeV that has a Maxwellian distribution, so it can be precisely described by:
T= 1.3 MeV. <ref > Leo</ref>
It is important to notice that 96.9% of all of Cf-252 decays are \alpha decays, and only 3.1 % occur through spontaneous fission. (considering the half life is 85.5 yeas for SF and 2.73 years for alpha decay).<ref > Reuss </ref>
- B- Industrial sources (installed or Laboratory sources)
The production the industrial neutron sources is by particle accelerator, Van de Graaff electrostatic accelerator, or by nuclear reactor. In case of using a particle accelerator, photon activation is one of the methods to produce this type of sources by carefully choosing an appropriate target.
When Van de Graaff accelerator is used, it projects a deuteron or a proton toward the target to produce neutrons, for example,
d ()n + 1.27 MeV
d ()n + 4.36 MeV
d ()n - 1.65 MeV <ref > Reuss </ref>
The nuclear reactor is rich environment for many radioactive isotopes that are produced by a neutron fission, a neutron activation, or other processes. For instance, .
Neutron energy: types and spectra
Neutron are classified into four types depending on their energy, each type has energy range which may change from one reference to another. The following table shows the types of neutrons, their energy range, and common detective materials.
| Neutron Type | Energy Range | Common detective material(s) |
| Ultracold Neutrons | less than 2*10^-7 | He-3 |
| Very Cold Neutrons | 2*10^-7eV =< K.E <= 5*10^-5 eV | He-3 |
| Cold Neutrons | 5*10^-5 eV =< K.E. =< 0.025 eV | He-3 |
| Thermal Neutrons | K.E ~_ 0.025 eV | Be-4, Li-6 |
| Epithermal Neutrons | 1 eV =< K.E =< 0.1 keV | |
| Intermediate Neutrons | 1 keV =< K.E =< 0.1 MeV | |
| Fast Neutrons | K.E > 0.1 MeV | U-238, Th-232 |
Neutron Induced Fission of U233 Cross Sections
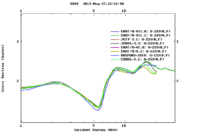
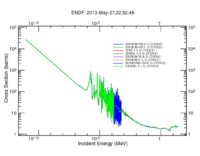
The definition of neutron induced fission cross section is the probability of occurrence of a fission interaction by an incident neutron that has a defined energy. Actinides and lanthanides generally have high fission cross sections compared to those of the rest of periodic table elements; for instance, U-235 has a high fission cross section for thermal neutrons, U-238 has a fission cross section for fast neutrons in barns, the same U-233 has a fission cross section in barns for almost all neutron energy range that starts with thermal neutron and ends with fast neutrons up to more than 30 MeV as shown in the following figure.
The interaction rate of the neutron with the fissionable material depends on the fission cross sections, microscopic and macroscopic fission cross sections, the microscopic cross section defined the effective area for a single nucleus for an interaction. The macroscopic cross section determines the probability of a neutron to interact when it travels a unit distance in a macroscopic material, its unit is (which is the reciprocal of mean free path), it is defined mathematically in terms of the microscopic cross section by:<ref name="Knief"> Knief R.A., (1992). Nuclear Engineering: Theory and Technology of Commercial Nuclear Power. 2nd ed. e.g. England: Hemisphere Publishing Corporation. </ref>:
Where is the macroscopic cross section and is the microscopic cross section , n is the material (target) atomic density .
When a neutron flux () interacts with a material of thickness x, the flux exponentially attenuates inside the material as shown by the formula:
so,
The microscopic cross section of the material is:
Where is the number incident particle per uni area per unit time, dN is the average number of particles per unit time that interacted per unit solid angle, and is the solid angle.
Since the cross section has an area unit (barn), some authors define this quantity as the area to which the particle is exposed to make an interaction. <ref name="Ahmed">Syed Ahmed, Physics and Engineering of Radiation Detection (Academic Press 2007) </ref> The cross section values are represented as a function of energy that gives the value of the cross section for each energy value and shows the resonance peaks. A theoretical description of the neutron fission cross section curve over all an energy range is not available, but statistically it is possible to evaluate the parameters for an assumption that describes part of the cross section curve within a certain error.<ref name="Hyde"/>
Neutron fission is one of the interactions that commonly takes place spontaneously or under certain experimental conditions. An incident neutron depositing sufficient energy in a nucleus to enable the nucleus to overcome the Coulomb barrier force, when the nucleus overcomes the barrier, it splits into lighter nuclei (fragments) and particles. The new products interact with the surrounding medium depending on their energy state, their half lives, and the type of the medium that contains the interaction.
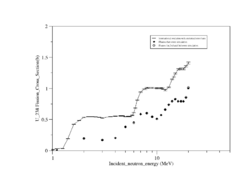
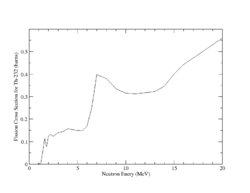
Both U-238 and Th-232 are members of the actinides group. They are characterized by relatively high neutron fission cross sections for fast neutrons with fission thresholds above the thermal neutron energy. The fission reaction in both elements is expected to eject 1-2 neutrons when the incident neutron energy is between 5-10 MeV as shown in Fig.9 and Fig.10.
 |
|
| Fig.9 U 238 fission cross section | Fig.10 Th 232 fission cross section <ref name="endf_Th_232_JEFF1.3">http://www.nndc.bnl.gov/exfor/servlet/E4sMakeE4 </ref> |
<references/>
Read more: http://www.americanessays.com/tool-box/apa-format-citation-generator/#ixzz2P8Ae4GVv