Difference between revisions of "Forest He-3 Tubes"
| Line 1: | Line 1: | ||
| − | |||
The neutron capture of He-3 may be represented by the reaction below | The neutron capture of He-3 may be represented by the reaction below | ||
<math>n + He^3 \rightarrow p + H^3</math> | <math>n + He^3 \rightarrow p + H^3</math> | ||
| − | About 764 keV of energy is liberated in this nuclear reaction and distributed between the final products according to their masses. Because the proton is | + | About 764 keV of energy is liberated in this nuclear reaction and distributed between the final products according to their masses. Because the proton is about a factor of 3 lighter than Tritium (H^3) , it will have more kinetic energy by about a factor of 3 (about 573 keV). This liberated proton can ionize other He-3 atoms via the reaction |
| − | |||
<math>p+He^3 \rightarrow p + He^3(+) + e^-</math> | <math>p+He^3 \rightarrow p + He^3(+) + e^-</math> | ||
| + | The same proton will ionize several He-3 atoms when dissipating the 573 keV kinetic energy. | ||
| + | |||
| + | The Tritium (H^3) can also ionize the gas but due to its higher mass it does not travel as far (shorter range) as the proton and makes a smaller contribution to the ionization signal. | ||
| + | |||
| + | reference: J. W. Leake, "Nuclear Instruments and Methods", Vol. 63, page 329, 1968). | ||
[[Image:He-3Tube_DetectorDrawing.jpg]] | [[Image:He-3Tube_DetectorDrawing.jpg]] | ||
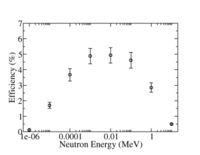
[[Image:He-3Tube_PPND_Efficiency.jpg | 200 px]] | [[Image:He-3Tube_PPND_Efficiency.jpg | 200 px]] | ||
Revision as of 09:50, 6 June 2008
The neutron capture of He-3 may be represented by the reaction below
About 764 keV of energy is liberated in this nuclear reaction and distributed between the final products according to their masses. Because the proton is about a factor of 3 lighter than Tritium (H^3) , it will have more kinetic energy by about a factor of 3 (about 573 keV). This liberated proton can ionize other He-3 atoms via the reaction
The same proton will ionize several He-3 atoms when dissipating the 573 keV kinetic energy.
The Tritium (H^3) can also ionize the gas but due to its higher mass it does not travel as far (shorter range) as the proton and makes a smaller contribution to the ionization signal.
reference: J. W. Leake, "Nuclear Instruments and Methods", Vol. 63, page 329, 1968).